31 Execution of Movement
Motor cortex
Once the plan for movement has been created, the primary motor cortex is responsible for the execution of that action. The primary motor cortex lies just anterior to the primary somatosensory cortex in the precentral gyrus located in the frontal lobe.
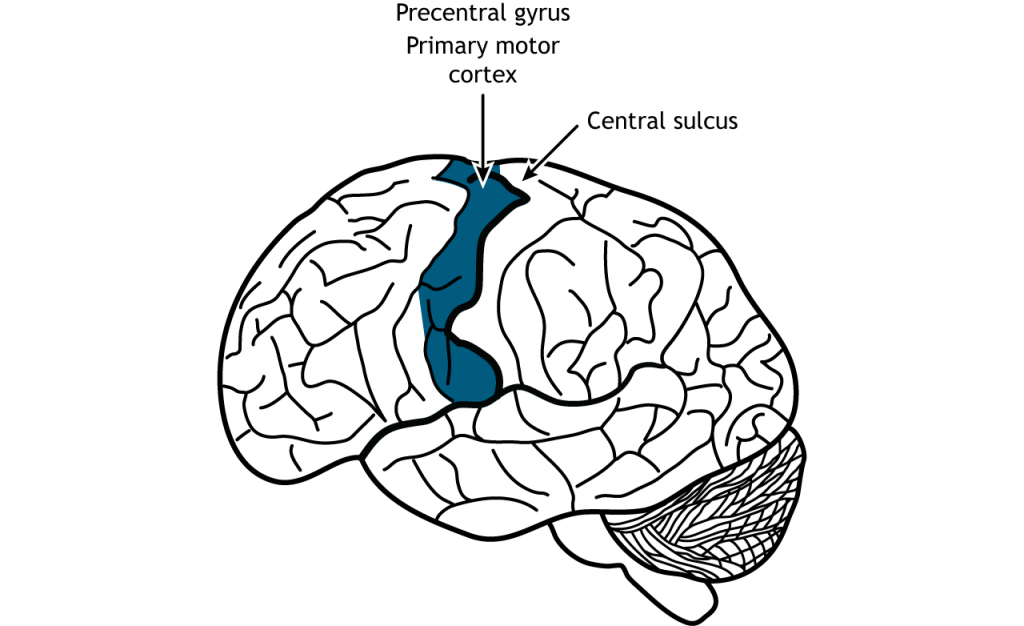
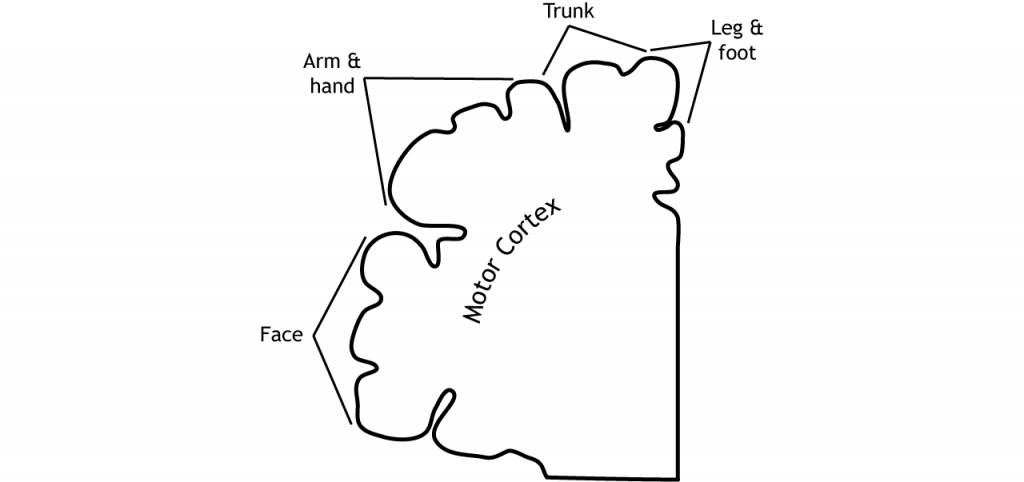
Like the somatosensory cortex, the motor cortex is organized by a somatotopic map. However, the motor cortex does not map onto the body in such an exact way as does the somatosensory system. It is believed that upper motor neurons in the motor cortex control multiple lower motor neurons in the spinal cord that innervate multiple muscles. This results in activation of an upper motor neuron causing excitation or inhibition in different neurons at once, indicating that the primary motor cortex is responsible for movements and not simply activation of one muscle. Stimulation of motor neurons in monkeys can lead to complex motions like bringing the hand to the mouth or moving into a defensive position (Graziano et al, 2005).
Population coding
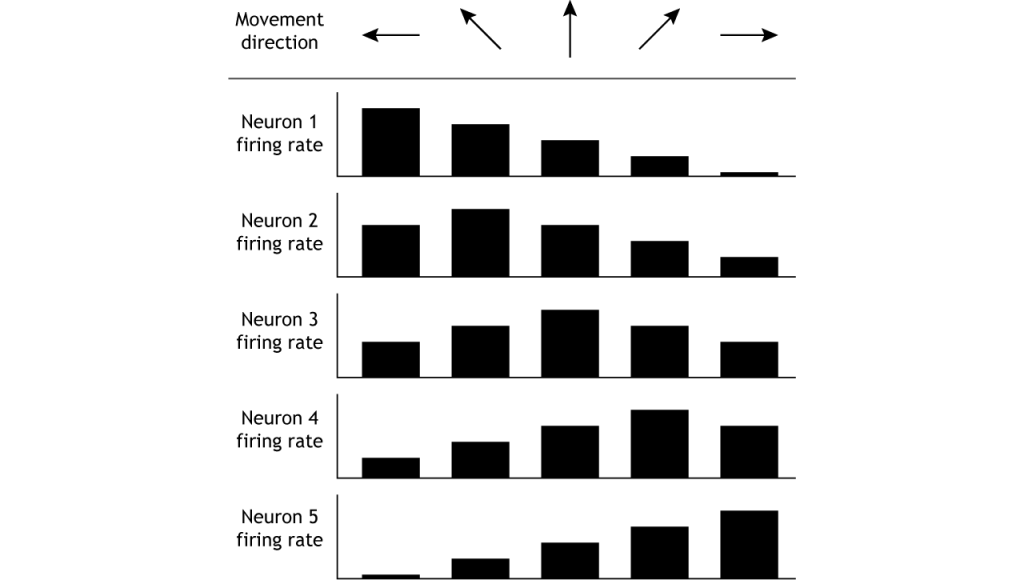
The motor cortex controls movement by using population coding mechanisms. Upper motor neurons are broadly tuned to a certain movement in a certain direction, meaning firing rate is highest when moving in one direction, but firing also occurs when moving in nearby directions. For example, when a monkey is trained to move its hand toward the left, neurons “tuned” toward left movement will be active immediately before and during the movement. Neurons tuned to other directions will also be active but at lower rates (Georgopoulos, et al, 1982). This means that the firing rate of one specific neuron does not give enough information to know direction of movement. It is the combined firing rates of an entire population of neurons that indicates direction.
Descending Spinal Tracts
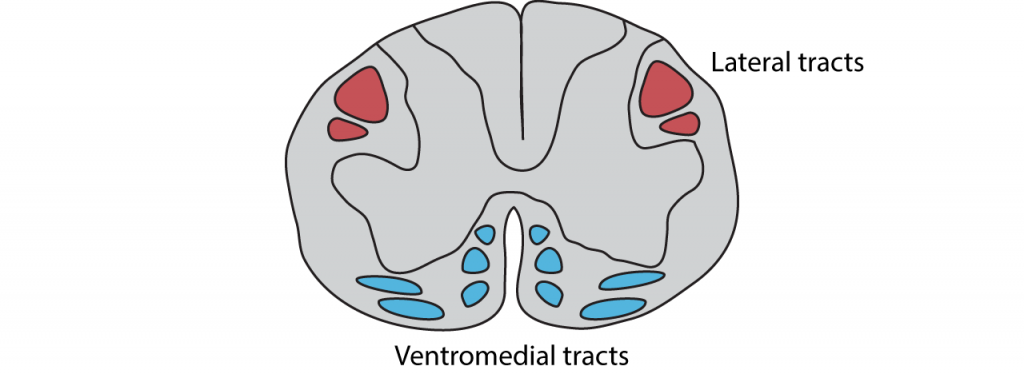
There are multiple descending tracts within the spinal cord that send information from the brain to the motor neurons in the ventral horn. The lateral tracts are responsible for carrying information about voluntary movement of the arms and legs. The ventromedial pathways are responsible for carrying information about posture and balance.
Lateral Tracts
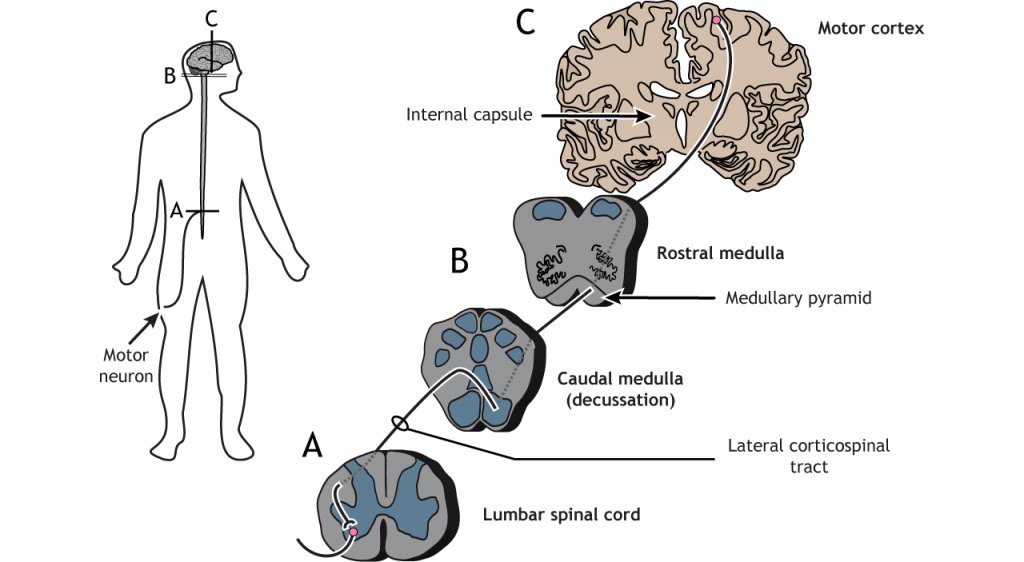
Coticospinal Tract
The largest of the lateral pathways is the corticospinal tract. This pathway sends information directly from the motor and premotor cortices down to the motor neurons in the spinal cord. Cortical axons travel through the brainstem and then cross the midline at the base of the medulla; like the somatosensory system, the right side of the cortex processes information for the left side of the body and vice versa. In the spinal cord, the axons travel through the lateral column and synapse in the ventral horn on motor neurons that typically innervate distal muscles.
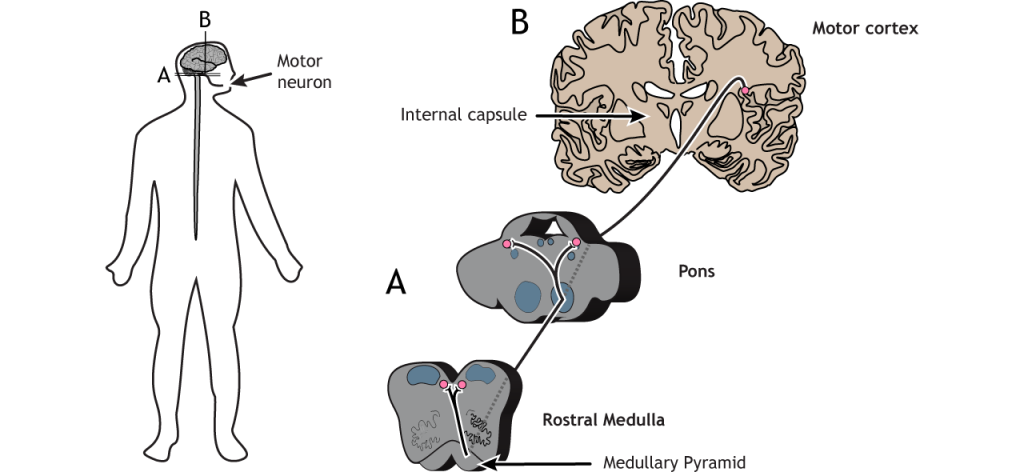
Corticobulbar Tract
The corticobulbar tract is another lateral tract and sends motor information to cranial nerves for motor control of the face. This path travels ipsilateral from the cortex into the brainstem where it branches off at the appropriate cranial nerve level in either the pons or the medulla and then innervates cranial nerve neurons bilaterally.
Ventromedial Tracts
There are four ventromedial pathways that travel in the spinal cord as well. These tracts begin in the brainstem and descend through the ventromedial columns. They receive input from motor areas of the cortex as well as integrating information from multiple sensory regions.
- The vestibulospinal tract is important for head balance as we move. This tract begins in the vestibular nucleus.
- The tectospinal tract is responsible for moving the head in response to visual stimuli. This tract begins in the superior colliculus.
- The two reticulospinal tracts play a role in managing anti-gravity reflexes needed for posture and standing. These tracts begin in the reticular formation.
Key Takeaways
- The motor cortex is located in the frontal lobe
- The motor map is not as detailed as the somatosensory homunculus
- The motor cortex uses population coding to encode direction of movement
- The lateral tracts carry information about voluntary movement of the arms and legs
- The ventromedial pathways carry information about posture and balance
Test Yourself!
Video Lecture
References
Georgopoulos AP, Kalaska JF, Caminiti R, Massey JT. J Neurosci. 1982 Nov;2(11):1527-37.
Graziano et al, 2005 J Neurophysiol 94: 4209-4223.

